The influence of seasonal changes in the physicochemical properties of fresh water on the rheological properties of hydraulic fracturing fluids (a case study of the Almetyevsk District, Republic of Tatarstan)
- 1 — Junior Researcher Almetyevsk State Technological University “Petroleum Higher School” ▪ Orcid
- 2 — Ph.D., Dr.Sci. Head of Department Almetyevsk State Technological University “Petroleum Higher School” ▪ Orcid
- 3 — Ph.D. Leading Expert on New Reagents PJSC Tatneft named after V.D.Shashin ▪ Orcid
- 4 — Engineer Almetyevsk State Technological University “Petroleum Higher School” ▪ Orcid
- 5 — Postgraduate Student Almetyevsk State Technological University “Petroleum Higher School” ▪ Orcid
- 6 — Chief Design Expert PJSC Tatneft named after V.D.Shashin ▪ Orcid
Abstract
The work investigates the influence of seasonal changes in the composition of fresh water on the rheological properties of guar‑borate hydraulic fracturing fluids. Between October 2024 and August 2025, monthly samples were taken from three sources in the Republic of Tatarstan, with analysis of pH (7.3-7.9), alkalinity (73-275 mg/l HCO-3), total hardness (180-520 mg/l Ca2⁺+ Mg2⁺), chlorides (42-284 mg/l), sulphates (61-146 mg/l), and iron ions (0.1-0.3 mg/l). Fracturing fluids were prepared using these waters and tested on a Brookfield PVS high‑pressure rheometer at 32 °C in accordance with ISO 3219:1993. The target viscosity range was approximately 400-700 mPa·s. We found that in water with high salinity (source N 2, average hardness ~419 mg/l, Cl–>180 mg/l), gel viscosity decreased by 10-15 %, the time to recover to operating viscosity increased, and breakdown accelerated, which raised the risk of premature proppant settling. Correlation analysis showed strong positive correlations between hardness, chlorides, sulphates, and alkalinity (r = 0.53-0.90) for “soft” sources N 1 and 3, whereas in the mineralized water of source N 2 these correlations are weakened (r = 0.17-0.51). The results demonstrate that the seasonal increase in salinity (winter‑spring period) significantly impairs the rheological stability of hydraulic fracturing fluids and emphasize the need for mandatory monitoring of water composition and adaptation of formulations during periods of peak salinity.
None
Introduction
Hydraulic fracturing (HF) is one of the most effective technologies for enhancing hydrocarbon recovery [1-3]. This technology requires significant operational and material resources, which depend on the properties of a specific geological target [4]. As more complex geological targets are developed, HF technology continues to evolve, leading to increased consumption of fresh water for preparing fracturing fluids, as well as higher demand for chemical reagents and process equipment [5-7].
Using fresh water as the base for fracturing process fluids is the global standard. This is due to its optimal properties, which ensure high efficiency and stability of the process without degrading the reservoir porosity and permeability or the rheological properties of the fluids. At the same time, using such water entails considerable costs [8], including transportation and time‑related expenses, especially during the winter and autumn periods. This encourages the search for alternative sources [9, 10], but in practice, fresh water remains the most preferred choice for HF operations [11-13].
Fresh water sources are typically natural water bodies. Such water is subject to seasonal fluctuations in physicochemical composition, which can significantly affect the operation efficiency. Water composition varies with the time of year, driven by a number of factors, including snowmelt, precipitation intensity, temperature fluctuations, and changes in the biological activity of water bodies [14-16].
It is known that water bodies exhibit seasonal variations in salinity and hardness [17, 18]. For example, in the Kuibyshev and Nizhnekamsk water bodies, salinity can increase significantly in winter and spring, while hardness may decrease in summer [19]. High water salinity can have a considerable impact on the rheological properties of polysaccharide gel, reducing its effective viscosity [20-22].
Seasonal changes in water temperature affect its physicochemical properties [23-25]. For instance, in summer, elevated temperatures can reduce salt solubility due to increased kinetic energy of water molecules, improved diffusion, and decreased solution viscosity. This may lead to increased water salinity and changes in its chemical composition. In spring, an increase in biological activity in water bodies is observed, causing higher levels of organic substances and changes in water pH. These alterations can affect the stability of HF fluids and require the use of additional stabilizing additives [26-29].
Considering the seasonal fluctuations in the composition of fresh water, it is necessary to conduct regular monitoring of its quality and adapt the composition of HF fluids depending on the current water properties. This will help maintain the efficiency of the operation and minimise risks associated with changes in the fracturing fluid properties.
The work analyses changes in the physicochemical properties of fresh water depending on the season and determines their impact on the rheological properties of fracturing fluids.
Methods
Fresh water from three regional sources used for preparing HF fluids in the Republic of Tatarstan was selected for the study. Water samples were collected monthly and subjected to physicochemical analysis using HACH test kits. The analysis determined the following parameters: pH, alkalinity by HCO3¯ , chloride Cl⁻ content, sulphate SO42¯ content, calcium Ca2+ and magnesium Mg2+ ions (total hardness), iron Fe2+ ions.
Rheological studies were conducted in accordance with the ISO 3219:1993 standard. Key rheological properties were determined via shear sensitivity tests (simulating fluid movement through production tubing followed by movement through the reservoir bed) and tests of the breakdown rate of the crosslinked system. The studies were carried out using Brookfield PVS high‑pressure rheometers at a temperature of 32 °C. To improve the research accuracy, shear sensitivity tests were performed twice for each sample. The main parameters determined in the rheological tests were the time required to restore dynamic viscosity after removing the shear load and the system breakdown rate.
Results of the physicochemical water analysis
The analysis results are presented in Tables 1-3. The study commenced in October 2024. Despite the fact that seasonal variations in water parameters from source N 1 are not large in magnitude, such fluctuations have a significant impact on the formation and stability of the guar‑borate gel. An increase in hardness and salinity by even 50-100 mg/l reduces the maximum viscosity by 10-15 % and accelerates breakdown. This is critical for efficient proppant placement and maintaining the fracture after HF.
Table 1
Results of the physicochemical analysis of fresh water from source N 1
|
Time from the first sample, days |
pH |
HCO3¯,mg/l |
Сl–, mg/l |
SO42¯,mg/l |
Mg2+, Ca2+, mg/l |
Fe2+, mg/l |
|
0 |
7.65 |
94.3 |
80.2 |
85.4 |
184.5 |
0.2 |
|
24 |
7.48 |
101.3 |
93.5 |
91.2 |
193.7 |
0.1 |
|
57 |
7.61 |
105.2 |
101.4 |
88.6 |
201.2 |
0.1 |
|
74 |
7.71 |
100.4 |
103.2 |
104.4 |
218.4 |
0.2 |
|
123 |
7.68 |
183 |
102.9 |
122.8 |
250.25 |
0.1 |
|
148 |
7.74 |
146.4 |
81.6 |
85.5 |
210.2 |
0.1 |
|
173 |
7.94 |
122 |
71 |
76.8 |
180.18 |
0.2 |
|
203 |
7.45 |
85.4 |
49.7 |
76.8 |
140.14 |
0.1 |
|
231 |
7.54 |
103.7 |
53.5 |
76.8 |
155.16 |
0.2 |
|
267 |
7.52 |
128.1 |
49.7 |
61.44 |
150.15 |
0.1 |
|
285 |
7.61 |
111.2 |
54.4 |
91.2 |
171.5 |
0.2 |
Table 2
Results of the physicochemical analysis of fresh water from source N 2
|
Time from the first sample, days |
pH |
HCO3¯,mg/l |
Сl–, mg/l |
SO42¯,mg/l |
Mg2+, Ca2+, mg/l |
Fe2+, mg/l |
|
0 |
7.62 |
208.4 |
201.8 |
114.4 |
400.4 |
0.1 |
|
24 |
7.71 |
201.6 |
205.7 |
95.4 |
394.2 |
0.1 |
|
57 |
7.78 |
193.2 |
195.4 |
114.4 |
511.4 |
0.2 |
|
74 |
7.82 |
224 |
213.6 |
98.6 |
496.5 |
0.1 |
|
123 |
7.42 |
274.6 |
282.22 |
145.9 |
520 |
0.1 |
|
148 |
8 |
244 |
284 |
84.48 |
400.4 |
0.1 |
|
173 |
7.66 |
198.24 |
181.1 |
107.5 |
395.4 |
0.1 |
|
203 |
7.9 |
224 |
191.7 |
92.16 |
305.4 |
0.1 |
|
231 |
7.61 |
244 |
213 |
115.2 |
384 |
0.2 |
|
267 |
7.45 |
231.8 |
209.45 |
122.88 |
395.39 |
0.1 |
|
285 |
7.66 |
213 |
201 |
105.4 |
401.5 |
0.1 |
Table 3
Results of the physicochemical analysis of fresh water from source N 3
|
Time from the first sample, days |
pH |
HCO3¯,mg/l |
Сl–, mg/l |
SO42¯,mg/l |
Mg2+, Ca2+, mg/l |
Fe2+, mg/l |
|
0 |
7.52 |
105.4 |
76.4 |
84.5 |
130.2 |
0.2 |
|
24 |
7.62 |
111.2 |
82.5 |
97.4 |
155.7 |
0.2 |
|
57 |
7.58 |
140.3 |
91.4 |
101.5 |
184.2 |
0.3 |
|
74 |
7.68 |
122.6 |
88.4 |
96.7 |
206.7 |
0.2 |
|
123 |
7.65 |
122.2 |
95.8 |
122.9 |
293 |
0.3 |
|
148 |
7.66 |
140.3 |
78.1 |
84.5 |
205.2 |
0.3 |
|
173 |
7.86 |
125 |
85 |
99.84 |
170.2 |
0.3 |
|
203 |
7.25 |
73.2 |
42 |
76.8 |
135.14 |
0.3 |
|
231 |
7.45 |
122 |
42.6 |
96 |
177.6 |
0.2 |
|
267 |
7.41 |
103.7 |
46.15 |
69.12 |
130.13 |
0.2 |
|
285 |
7.45 |
110.5 |
52.1 |
76.8 |
177.6 |
0.3 |
Source N 1 (Kama water) – water sampled from the main channel of the Kama River in its upper reaches. The water is predominantly hydrocarbonate‑calcium type, with low salinity; hardness is moderate (approximately 180-200 mg/l by Ca2++Mg2+). It is characterized by pronounced seasonal variability (spring flood period → decrease in salinity; summer – autumn period → increase due to evaporation and runoff).
Source N 2 (Karabash Reservoir) – a surface storage-type water body, formed by river inflow and atmospheric recharge. The water is chloride-sulphate-calcium-magnesium type, mineralized, with elevated hardness (average 420 mg/l). It has a relatively stable composition throughout the year but inherently contains high concentrations of Cl⁻ and SO42¯.
Source N 3 (Kama water) – water sampled from the Kama River further downstream (at a different section). The composition is similar to that of source N 1 (hydrocarbonate-calcium type, low salinity). Seasonal dynamics is observed, associated with surface runoff and the hydrological regime of the river.
The physicochemical water analysis showed that water from source N 2 is notably characterized by high salinity. For instance, the average water hardness in source N 2 over the course of a year is 419 mg/l, whereas in sources N 1 and N 3 this parameter ranges from 180 to 190 mg/l. Maximum water hardness values (approximately 520 mg/l) in source N 2 are observed during the transitional winter-spring season. It can be noted that in general water from sources N 1 and N 3 is similar in composition. The pH of water from all sources remains within the range of 7.3-7.9 throughout the year.
The pH and iron content can be considered stable parameters, as they exhibit only minor variations. The coefficient of variation is approximately 5 % for pH and 30 % for iron (pH varies within 0.5, while iron content varies within 0.1-0.2 mg/l). Variable parameters include alkalinity, hardness, chloride and sulphate content, as variations in these indicators are considerable – especially in water from source N 2. Fig.1 presents the dynamics of changes in the content of various components in the water depending on the time of year.
The research shows that the environment at all water sources remains slightly alkaline throughout the year; however, pH values slightly decreased in summer (Fig.1, e). This may be associated with increased biological activity [30]. The dynamics of pH changes generally correlate with alkalinity (Fig.1, a), particularly noticeable in source N 3. When alkalinity decreases, the buffering capacity of the water drops, which may lead to a slight decrease in pH.
Alkalinity increases in spring and decreases in summer. For example, in source N 1, alkalinity rises from 105.2 mg/l in February to 183 mg/l in April, then drops by half by July and rises again in autumn. A similar result is shown by the physicochemical analysis of source N 3. Source N 2 also exhibits maximum alkalinity in spring; however, summer values remain relatively high and do not show the same sharp decline as in the other sources. The maximum values of HCO3¯ may be associated with the influx of meltwater, which leaches carbonate compounds from the soil [31].
Total hardness (Fig.1, b) reaches maximum values for all sources at the end of winter and beginning of spring, while the water becomes significantly softer in summer. Water can be considered soft if it falls within the range up to 75 mg/l as CaCO₃, and of medium hardness up to 150-160 mg/l. Source N 2 is characterized by higher hardness throughout the year. The summer decrease in hardness may be related to the precipitation of carbonates when the water is heated, as well as dilution by rainfall.
The chloride ion content (Fig.1, c) follows a similar pattern. In the less mineralized sources N 1 and N 3, chloride levels are highest during the cold period and decrease in summer. An increase is then observed by autumn. In source N 2, chloride levels remain consistently high compared to the other sources. Moreover, the absolute level is two to three times higher than in the other sources, which may indicate the presence of mineralized groundwater.
The concentration of sulphate ions (SO42¯) generally follows a pattern similar to that of chlorides and hardness (Fig.1, d). All sources exhibit a spring maximum and a significant decrease by the end of summer. In source N 1, sulphate content increases from 85 mg/l in winter to 122.8 mg/l in April, then drops to 61 mg/l in September (a twofold decrease). Source N 3 shows a maximum value in April and a minimum in September. In source N 2, a peak of 145.9 mg/l in April is followed by a drop to 84.5 mg/l in May, which may be associated with intense precipitation. Subsequent summer values for source N 2 range between 92 and 115 mg/l, rising to 122 mg/l by autumn.
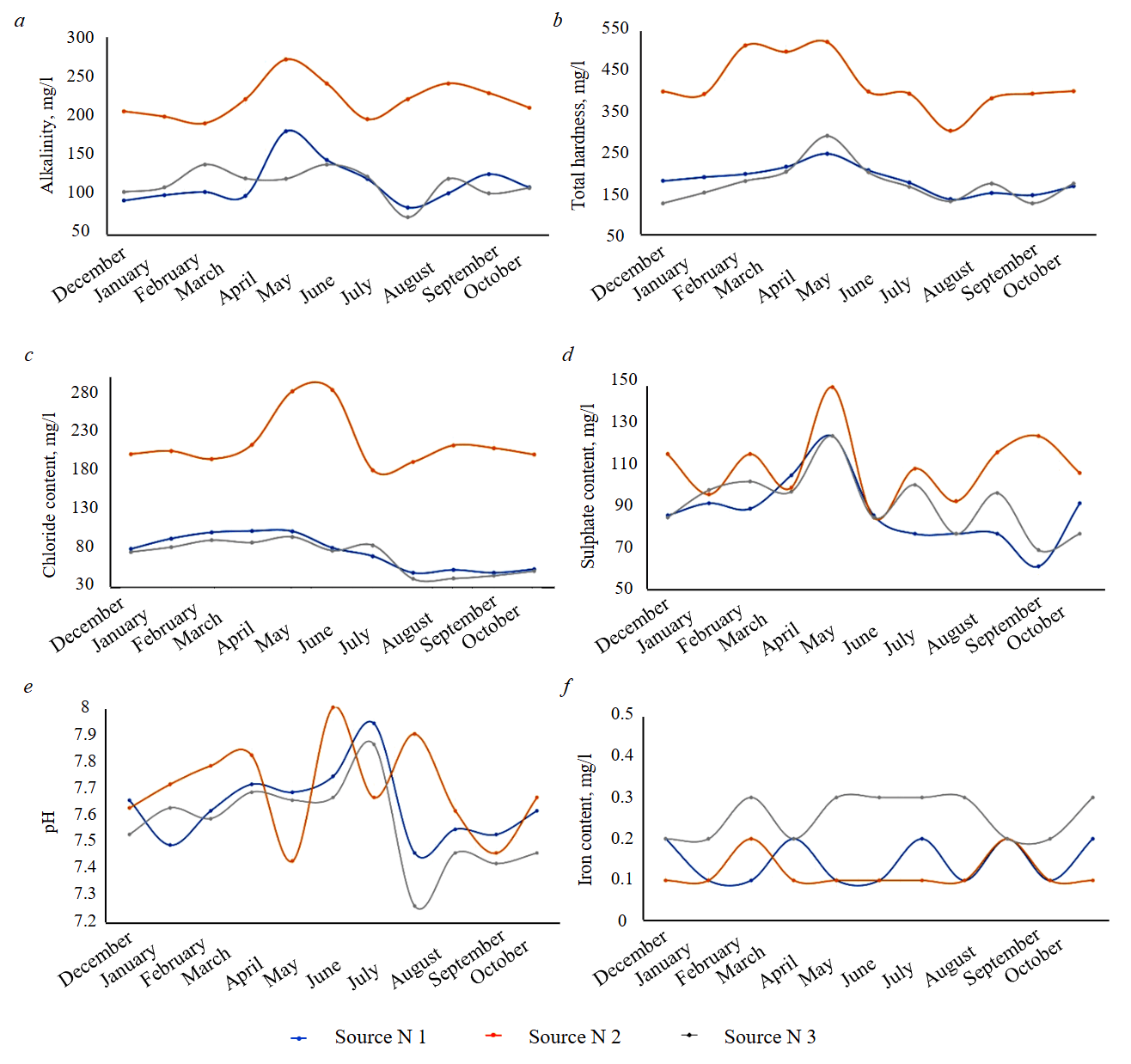
Fig.1. Dynamics of changes in the physicochemical composition of the studied water: a – alkalinity (HCO3¯); b – total hardness (Ca2+, Mg2+); c – chlorides (Cl–); d – sulphates (SO42¯); e – pH; f – iron (Fe2+)
The dissolved iron content (Fig.1, f) in the studied waters is relatively low and varies slightly throughout the year. In sources N 1 and N 2, iron levels remained stable at 0.1-0.2 mg/l, with no pronounced seasonality. Source N 3 showed slightly higher concentrations in summer (0.3 mg/l from February to July and 0.2 mg/l in autumn and winter). Nevertheless, the absolute values of Fe2+ are very low and are unlikely to cause immediate process constrains. No significant correlations were found between iron content and other parameters – i.e., fluctuations in Fe2+ occur independently of the total water salinity.
Correlation analysis of parameters
To identify relationships between parameters, the Pearson correlation coefficient [32] was estimated for the entire sample and for each source individually (Fig.2). The p-value is provided as a criterion for correlation significance. The general trend is as follows: in sources with softer water (N 1 and N 3), most inorganic components change almost synchronously. In contrast, in the mineralized water of source N 2, the relationships are weakened.
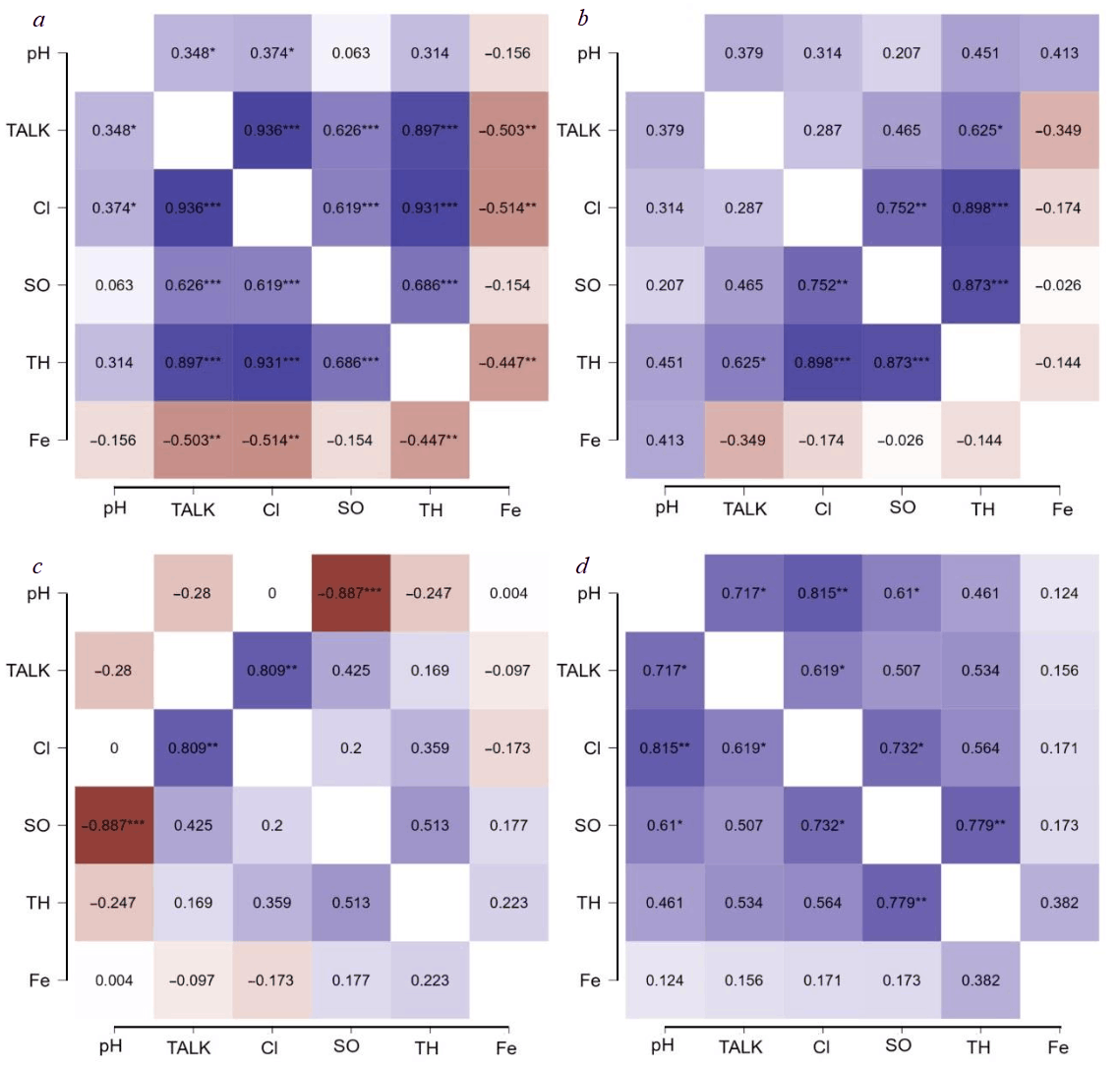
Fig.2. Pearson correlation for water parameters: a – for the entire sample; b – water source N 1; c – water source N 2; d – water source N 3
* p < 0.05; ** p < 0.01; *** p < 0.001; TALK – total alkalinity
Sources N 1 and N 3 show a moderate positive correlation between hardness and alkalinity (r = 0.53-0.62). This indicates that the main contribution to total hardness comes from bicarbonate salts of calcium and magnesium. In source N 2, this relationship is weak (r = 0.17), which may suggest a more complex composition. Most likely, hardness in this source is increased by other anions (sulphates, chlorides), while the proportion of bicarbonates is lower.
Water from sources N 1 and N 3 shows a very high correlation of hardness with chlorides (r = 0.90) and sulphates (r = 0.78-0.87). That is, when total salinity increases, the content of Ca2+ and Mg2+, Cl⁻ and SO42¯ ions simultaneously rises. This means that all dissolved salts behave as a single unit and depend on the degree of water dilution. In source N 2, these correlations are lower (r = 0.36 for Cl⁻, r = 0.51 for SO42¯). This shows that changes in hardness are not fully determined by changes in chloride and sulphate levels. For example, in spring, hardness in source N 2 increased significantly (exceeding 500 mg/l), while chloride level did not rise as substantially – likely due to the influx of soft water.
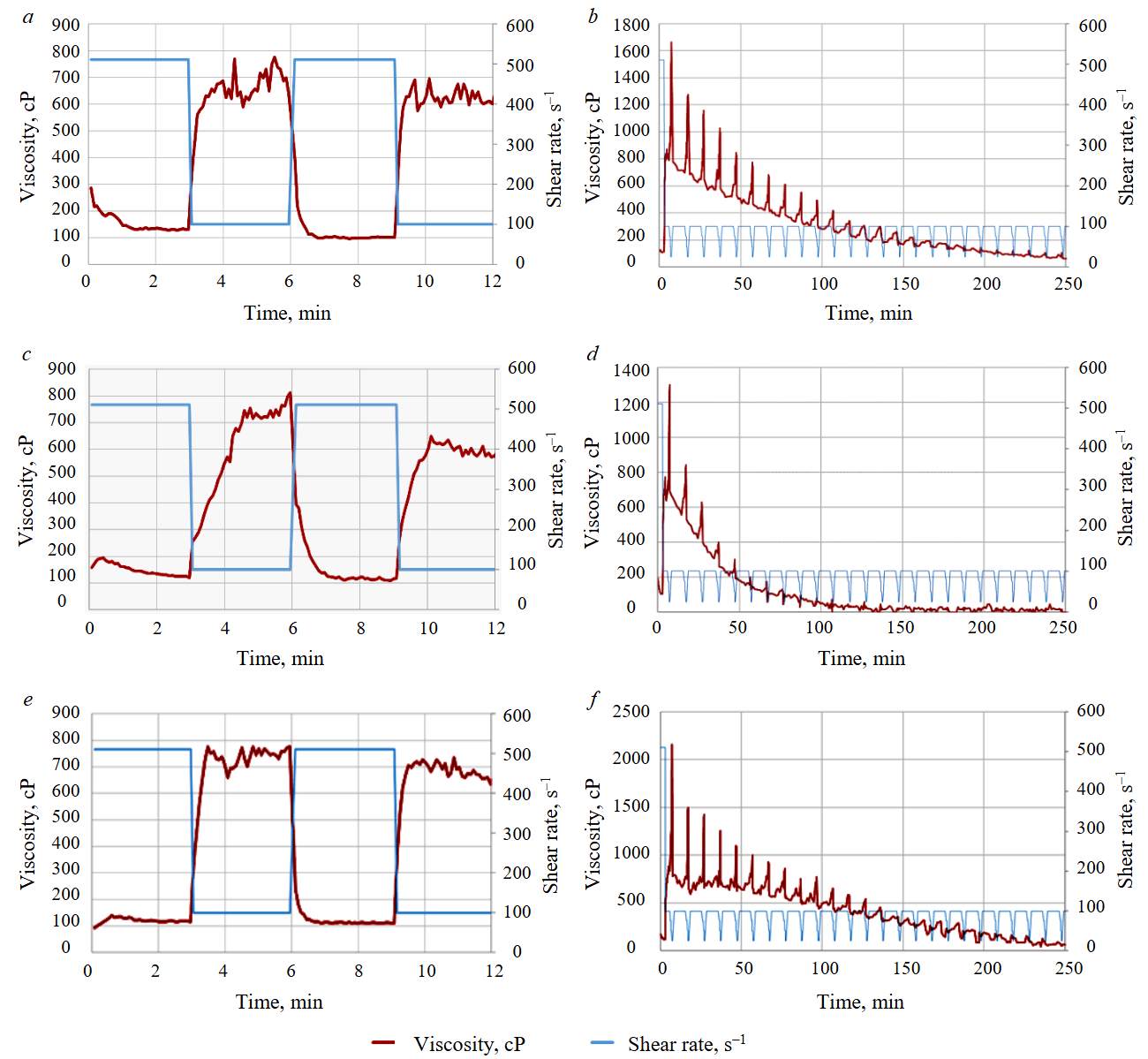
Fig.3. Rheological tests of water-based fracturing fluid from sources N 1 (a, b); N 2 (c, d); N 3 (e, f); for shear sensitivity (a, c, e); for decomposition rate (b, d, f)
In sources N 1 and N 3, chloride and sulphate levels change synchronously (r = 0.75), whereas in source N 2 there is virtually no correlation between them (r = 0.20). This implies differences in the origin of these salts. Possibly, in source N 2, part of the sulphates comes from a separate source (e.g., from gypsum strata) and may precipitate independently of chlorides.
A positive correlation between pH and alkalinity was expectedly observed, particularly strong for source N 3 (r = 0.72). This means that at high alkalinity, the water is better buffered and maintains a slightly higher pH. In source N 2, such a relationship is not observed, which may be due to the influence of other factors.
No significant relationship was found between pH and Fe2+ content (r < 0.15 across all sources). This may be related to the fact that iron is present in too low concentrations.
The correlation analysis confirms that for relatively clean fresh water (sources N 1 and N 3), total salinity acts as the main factor jointly influencing hardness, chlorides, sulphates, and partially alkalinity. In contrast, the water composition in source N 2 is more mixed: it shows relatively high concentrations of salts of various origins compared to the other sources. Therefore, linear dependencies between individual ions are not so obvious.
Influence of seasonal water composition on the hydraulic fracturing fluid properties
Rheological studies were carried out in the following sequence:
- a linear gel (water with a guar-based gelling agent) was prepared using a Waring laboratory blender by hydration for 20 min;
- a range of chemical reagents was added to the linear gel, including standard additives for fracturing fluids: demulsifier, clay stabilizer, breaker, breaker activator;
- the gel was crosslinked using a special additive, after which tests were conducted on a Brookfield PVS high-pressure rheometer.
The studies included shear sensitivity tests – simulating fluid movement through production tubing followed by release into the reservoir bed; breakdown rate tests – to determine behaviour and consistency indicators. The required range of dynamic viscosity for a standard guar-borate fracturing fluid is approximately 400-700 mPa∙s.
Results of rheological studies show that fluctuations in the physicochemical composition of water throughout the year significantly affect the rheology of crosslinked guar‑borate fracturing fluids used in HF (examples in Fig.3). Based on the results of shear sensitivity and breakdown rate tests, the following was observed:
- Using gel prepared with water of elevated salinity from source N 2 leads to a decrease in the initial viscosity of the resulting gel. Most likely, high hardness and chloride content hinder full hydration of the guar base and may disrupt the action of the crosslinking agent. As a result, the obtained crosslinked gel has lower strength between bonds. Compared to soft water (sources N 1 and N 3), gels prepared with hard water exhibit 10-15 % lower maximum viscosity and are presumably less effective at proppant retention.
- Gels prepared with relatively soft water show longer viscosity retention. Mineralized water leads to accelerated gel breakdown. Thus, in harder water, gel viscosity is lower by the time of injection into the formation, which creates risks of premature proppant settling.
Seasonal changes in water composition also affect the oxidative breaker performance. In highly mineralized water from source N 2, higher levels of impurities were detected, which presumably accelerated the breakdown of peroxide compounds. As a result, cases of accelerated breakdown were observed. Consequently, the same fracturing fluid formulation yielded different breakdown times depending on water properties.
Results
Changes in the parameters of the studied water (alkalinity, hardness, chloride, iron, and sulphate content) are seasonal in nature. The conducted monitoring showed that even moderate seasonal fluctuations in freshwater parameters (pH 7.3-7.9, alkalinity 73-275 mg/l, hardness 180-520 mg/l, Cl⁻ 42-284 mg/l, SO42¯ 61-146 mg/l, Fe2+ 0.1-0.3 mg/l) can significantly alter the rheological properties of guar‑borate HF fluids. When hardness values exceed 300 mg/l, chloride content is over 180 mg/l, and sulphate levels are above 100 mg/l, we observed a decrease in gel viscosity by 10-15 %, an increase in the time required to recover working viscosity, and accelerated breakdown. This does not meet the optimal requirements for water used in HF (hardness 200-250 mg/l, Cl⁻ < 150-180 mg/l, Fe2+ < 0.1 mg/l).
Comparison of the three sources showed noticeable differences in average composition. Source N 2 has significantly higher salinity. When planning HF operations, it is necessary to consider that using water of different origins may require different approaches. Less mineralized water is more favourable for preparing HF fluids, whereas more saline water may require special preparation measures or may even be unsuitable for HF.
The correlation analysis showed that the level of total water hardness can be significantly influenced by various ionic components. In low‑salinity waters (sources N 1 and N 3), strong positive correlations were observed between total hardness and chloride content (r = 0.90), sulphate content (r = 0.78-0.87), alkalinity (r = 0.53-0.62). This indicates a unified mechanism of water composition changes due to dilution or concentration of solutions. At the same time, in more mineralized water (source N 2), the relationships between these components are weakened (r = 0.17-0.51). This suggests a complex structure of the salt composition and the influence of additional factors. These data confirm that hardness cannot be considered as an independent parameter – it is a resulting indicator of complex seasonal processes occurring in water bodies.
Periods of increased water salinity mainly coincide with deterioration of the rheological properties of crosslinked gels. Their viscosity decreases and breakdown accelerates. Without adaptation of the formulation, this may lead to unpredictable behaviour of the HF fluid, for example, to premature proppant settling.
To ensure the stability of HF operations, it is necessary to conduct regular chemical water analysis and adjust the fracturing fluid composition according to seasonal conditions. In water with the highest hardness, it may be necessary to increase the loading of the gelling agent (to compensate for viscosity loss) or to use stabilizing additives.
The analysis results confirm the need for an adaptive approach to the development and selection of HF fluids. Instead of a universal formulation, it is advisable to have a set of corrective measures for different water compositions. This ensures maintaining the optimal gel viscosity and controlled rate of its breakdown under any seasonal variations in water quality.
Conclusion
The study confirms the significant influence of seasonal changes in the physicochemical composition of fresh water on the rheological properties of fluids used in hydraulic fracturing procedure. We found that the most unfavourable conditions for the formation of stable crosslinked gels occur in the winter‑spring period, when water salinity reaches its maximum values. This leads to a decrease in gel viscosity and accelerated gel breakdown, increasing the risks of ineffective proppant placement and reduced fracture conductivity.
Thus, to ensure high efficiency and reliability of HF operations, it is necessary to implement an adaptive approach to selecting process fluid formulations. This approach should include mandatory monitoring of water sources and seasonal adjustment of the composition. This will significantly increase the sustainability of hydraulic fracturing processes, minimize costs associated with unsuccessful injections, and ensure more stable hydrocarbon production.
References
- Zhiqiang Xie, Dongya Han, Jiangteng Li, Kaihui Li. A State-of-the-Art Review of Hydraulic Fracturing in Geothermal Systems. Sustainability. 2024. Vol. 16. Iss. 24. N 11087. DOI: 10.3390/su162411087
- Hui Gao, Xiaohang Li, Teng Li et al. Characteristics of Oil Production by Fracturing Fluid Additive-Assisted Displacement in Tight Oil Reservoirs. Energy & Fuels. 2024. Vol. 38. Iss. 18, p. 17541-17553. DOI: 10.1021/acs.energyfuels.4c02634
- Hao Yu, WenLong Xu, Bo Li et al. Hydraulic Fracturing and Enhanced Recovery in Shale Reservoirs: Theoretical Analysis to Engineering Applications. Energy & Fuels. 2023. Vol. 37. Iss. 14, p. 9956-9997. DOI: 10.1021/acs.energyfuels.3c01029
- Sultanov S.K., Mukhametshin V.Sh., Stabinskas A.P. et al. Study of the possibility of using high mineralization water for hydraulic fracturing. Journal of Mining Institute. 2024. Vol. 270, p. 950-962.
- Fokker P.A., Borello E.S., Verga F., Viberti D. Harmonic pulse testing for well performance monitoring. Journal of Petroleum Science and Engineering. 2018. Vol. 162, p. 446-459. DOI: 10.1016/j.petrol.2017.12.053
- Khan H.J., Spielman-Sun E., Jew A.D. et al. A Critical Review of the Physicochemical Impacts of Water Chemistry on Shale in Hydraulic Fracturing Systems. Environmental Science & Technology. 2021. Vol. 55. Iss. 3, p. 1377-1394. DOI: 10.1021/acs.est.0c04901
- Khuzin R.R., Andreev V.E., Mukhametshin V.V. et al. Influence of hydraulic compression on porosity and permeability properties of reservoirs. Journal of Mining Institute. 2021. Vol. 251, p. 688-697. DOI: 10.31897/PMI.2021.5.8
- Almuhametova E.M., Gutorov A.Yu., Yumadilov D.B. et al. Reducing the cost of hydraulic fracturing. Petroleum Engineering. 2024. Vol. 22. N 5, p. 103-112 (in Russian). DOI: 10.17122/ngdelo-2024-5-103-112
- Jiarun Zhou, Shibin Wang, Ruoyu Yang et al. A Study on a Novel Hydrophobic Associative Polymer Thickener for High-Salinity Water in Hydraulic Fracturing. ChemistrySelect. 2025. Vol. 10. Iss. 20. N e01613. DOI: 10.1002/slct.202501613
- Rodriguez A.Z., Huiyao Wang, Lei Hu et al. Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media. Water. 2020. Vol. 12. Iss. 3. N 770. DOI: 10.3390/w12030770
- Zang A., Hofmann H., Yinlin Ji et al. How rock hydraulic fatigue methods from mining and petroleum industry assist in unlocking deep heat for a clean energy future. Renewable and Sustainable Energy Reviews. 2025. Vol. 217. N 115683. DOI: 10.1016/j.rser.2025.115683
- Vylomov D.D., Shulgin P.A., Shakirov R.R., Snohin A.A. Selection of an alternative hydraulic fracturing fluid in the conditions of a low permeability gas reservoir turonian. Exposition Oil Gas. 2023. N 2 (95), p. 56-61 (in Russian). DOI: 10.24412/2076-6785-2023-2-56-61
- Nasybullin A.V., Sadreeva R.Kh., Burlutsky E.A. Determination of the efficiency of clay stabilizer component of fracturing fluid through coreflood experiments with terrigenous clay reservoir rock samples. Neftyanaya provintsiya. 2023. N 2 (34), p. 208-226 (in Russian). DOI: 10.25689/NP.2023.2.208-226
- Musin R.Kh., Galieva A.R., Khamitov A.D. Transformation of fresh groundwater composition over time in the Republic of Tatarstan. Uchenye zapiski Kazanskogo universiteta. Seriya Estestvennye nauki. 2023. Vol. 165. N 3, p. 427-446 (in Russian). DOI: 10.26907/2542-064X.2023.3.427-446
- Rakhimov I.I., Zainullin M.A., Ignashev N.E. Natural and technogenic water bodies as reserves for the conservation of biological diversity in the Republic of Tatarstan: problem statement and analysis. The Academic Journal of Moscow City University. Series “Natural Sciences”. 2023. N 2 (50), p. 21-36 (in Russian). DOI: 10.25688/2076-9091.2023.50.2.02
- Musin R.Kh., Kalkamanova Z.G. Geochemistry of fresh groundwater in regions with oil technogenesis (on the example of Tatarstan). Georesources, Geoenergetics, Geopolitics. 2015. N 2 (12), p. 14 (in Russian). DOI: 10.29222/ipng.2078-5712.2015-12.art12
- Musin R.Kh., Galieva A.R., Kudbanov T.G. et al. Soil influence on the chemical composition of ground waters in the republic of Tatarstan. Proceedings of higher educational establishments. Geology and Exploration. 2020. Vol. 63. N 1, p. 90-99 (in Russian). DOI: 10.32454/0016-7762-2020-63-1-90-99
- Korcheva E.S., Stepanova S.V., Shaikhiev I.G. Assessment of the Kazanka River water quality. Bulletin of the Technological University. 2016. Vol. 19. N 20, p. 186-189 (in Russian).
- Milke J., Gałczyńska M., Wróbel J. The Importance of Biological and Ecological Properties of Phragmites Australis (Cav.) Trin. Ex Steud., in Phytoremendiation of Aquatic Ecosystems – The Review. Water. 2020. Vol. 12. Iss. 6. N 1770. DOI: 10.3390/w12061770
- Mingazova N.M., Nabeeva E.G., Cheban E.Yu. et al. Water quality assessment of Kuibyshev reservoir in the Tatarstan Republic according to the expedition “Floating University”. Problemy ehkologii Volzhskogo basseina: Trudy 8-i vserossiiskoi nauchnoi konferentsii, 21-22 noyabrya 2023, Nizhnii Novgorod, Rossiya. Nizhnii Novgorod: Volzhskii gosudarstvennyi universitet vodnogo transporta, 2023. Iss. 6, p. 5 (in Russian).
- Leiming Li, Al-Muntasheri G.A., Feng Liang. A review of crosslinked fracturing fluids prepared with produced water. Petroleum. 2016. Vol. 2. Iss. 4, p. 313-323. DOI: 10.1016/j.petlm.2016.10.001
- Das P., Konale S., Kothamasu R. Effect of Salt Concentration on Base-gel Viscosity of Different Polymers used in Stimulation Fluid Systems. SPE/EAGE European Unconventional Resources Conference and Exhibition, 25-27 February 2014, Vienna, Austria. OnePetro, 2014. N SPE-167786-MS. DOI: 10.2118/167786-MS
- Earnden L., Laredo T., Marangoni A.G. et al. Modulation of the Viscosity of Guar-Based Fracking Fluids Using Salts. Energy & Fuels. 2021. Vol. 35. Iss. 19, p. 16007-16019. DOI: 10.1021/acs.energyfuels.1c02835
- Salnikova Yu.I. Results of studies on the compatibility of formation and injected water in hydrocarbon fields in Western Siberia. Advances in current natural sciences. 2024. N 2, p. 44-53 (in Russian). DOI: 10.17513/use.38217
- Shvydkyi V.O., Dubovik A.S., Kozlov M.V. et al. The Effect of the Physicochemical Properties and the Composition of Dubna River Water on the State of the Processes of Lipid Peroxidation in Biological Systems. Water Resources. 2024. Vol. 51. N 4, p. 525-533. DOI: 10.1134/S0097807824700933
- Kilyanov M.Yu., Igrevsky L.V., Khafizov S.F. et al. Efficiency improvement of Bactericides and Biostats Application when Using Seawater in a Reservoir Pressure Maintenance System. Kazakhstan journal for oil & gas industry. 2023. Vol. 5. N 3, p. 59-70 (in Russian). DOI: 10.54859/kjogi108636
- Pereira G.F., Rosin T.R., Braga B. et al. Evaluation of biofilm inhibition and detachment by commercial biocides in sulfate-reducing bacteria consortia from oil fields. Journal of Water Process Engineering. 2024. Vol. 63. N 105547. DOI: 10.1016/j.jwpe.2024.105547
- Buldakova N.S., Novikova N.V., Fakhrieva G.V. et al. Formation of approaches to the selection of bactericides to suppress the vital activity of sulphate-reducing bacteria. Oilfield engineering. 2020. N 6 (618), p. 68-72 (in Russian). DOI: 10.30713/0207-2351-2020-6(618)-68-72
- Mammedov K.A., Hamidova N.S. Application of Integrated Activity Bactericide for Corrosion Protection of Oilfield Equipment and Pipelines. Oil and Gas Territory. 2018. N 3, p. 20-25 (in Russian).
- Arroyo J.I., Díez B., Kempes C.P. et al. A general theory for temperature dependence in biology. PNAS. 2022. Vol. 119. N 30. N e2119872119. DOI: 10.1073/pnas.2119872119
- Mushaeva T.I., Demidov V.V. Regularities of formation and erosion processes during spring snowmelt on the territory of agrolandscape and their impact on the quality of river water. Live and bio-abiotic systems. 2015. N 11, p. 9 (in Russian).
- Okwonu F.Z., Chiyeaka O.M., Ahad N.A., Sharipov O. Robust Pearson correlation coefficient for imbalanced sample size and high dimensional data set. Science World Journal. 2025. Vol. 20. N 1, p. 131-136. DOI: 10.4314/swj.v20i1.17
